OvirusTdb - A resource to explore the therapeutic potential of oncolytic viruses
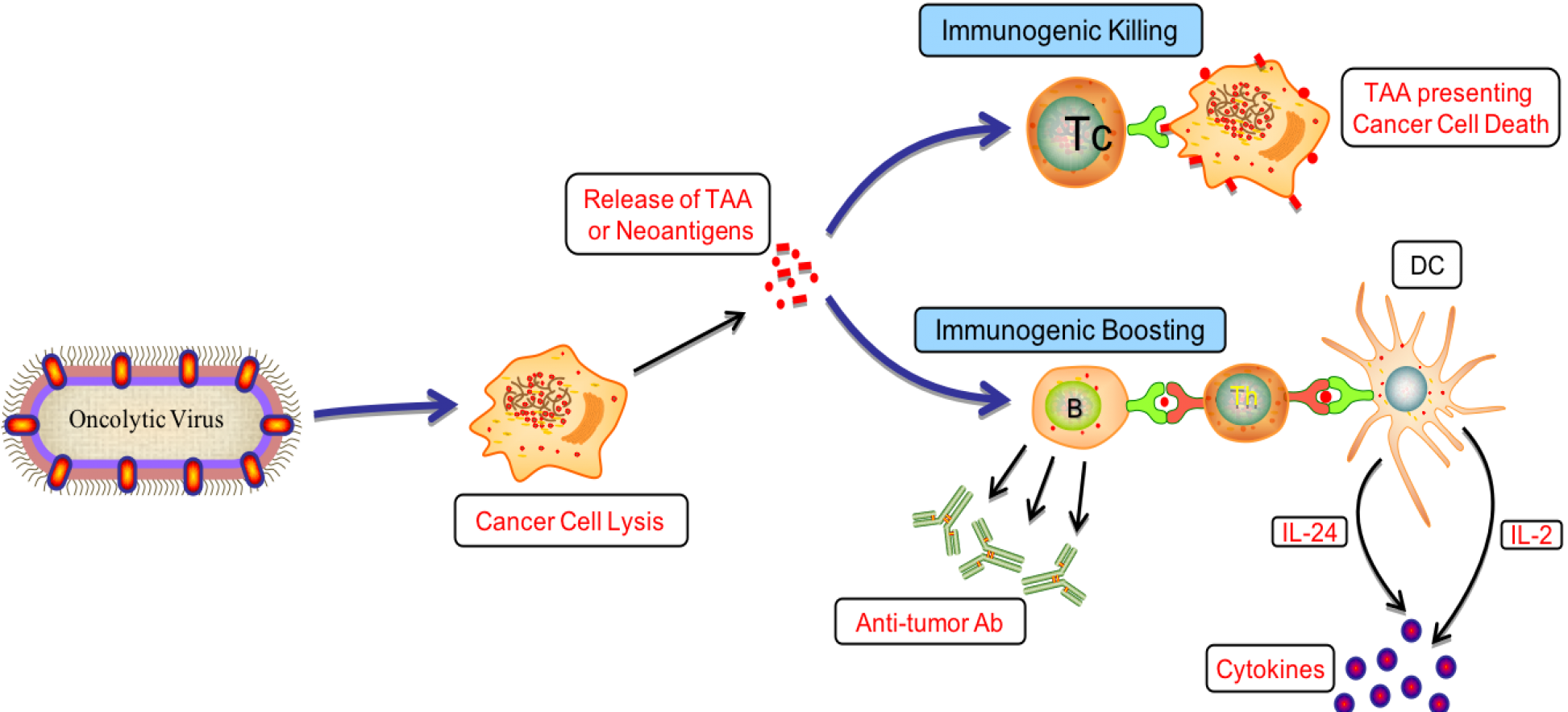
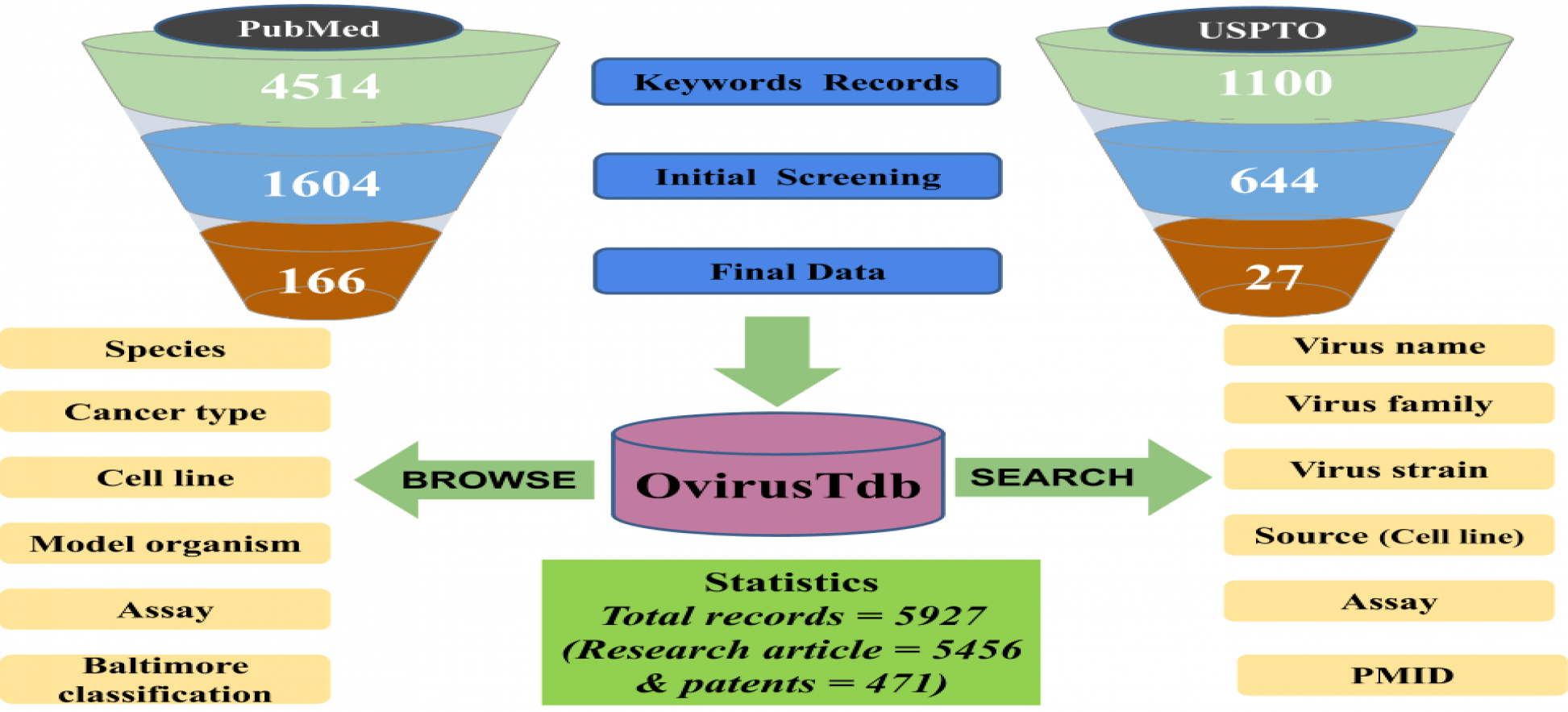
Cancer is the leading cause of death worldwide. According to GLOBOCAN database of world health organization (WHO) report 2018, the global cancer burden rise to 18.1 million new cases and 9.6 million deaths due to cancer. Scientists and researchers worldwide try to discover new molecules and treatment strategies to combat this deadly disease. Drugs such as paclitaxel, vincristine, vinblastine, docetaxel, NT-219 and several treatment strategies such as kinase inhibitor, mitotic disruptor, HDAC inhibitors, radiotherapy, etc. currently used to treat cancer. The limitation associated with current cancer treatment is that it is of long duration and increasing resistance of tumor cells towards chemotherapy along with several side effects. Cancer immunotherapy is the modern strategy for cancer treatment where some are approved such as interleukin-2 (IL) based therapy and several thousand molecules in clinical trials. Oncolytic viruses specifically spread in tumor cells without affecting normal cells can aid in cancer immunotherapy. OvirusTdb is a resource to explore the therapeutic potential of oncolytic viruses. Data has been curated from US patents and PubMed articles. It stores and manages information on 24 viruses, 427 cell lines, 22 model organisms and include 5927 records of oncolytic viruses.
References (If you are using above please cite following articles)
- Lathwal A., Kumar R., and Raghava GPS (2020) OvirusTdb: A database of oncolytic viruses for the advancement of therapeutics in cancer. Virology, doi.org/10.1016/j.virol.2020.05.016.
- Lathwal A, Kumar R, Raghava GPS (2020) Computer-aided designing of oncolytic viruses for overcoming translational challenges of cancer immunotherapy. Drug Discov Today. 2020;S1359-6446(20)30156-2.