A web server for predicting S-adenosyl-L-methionine binding sites
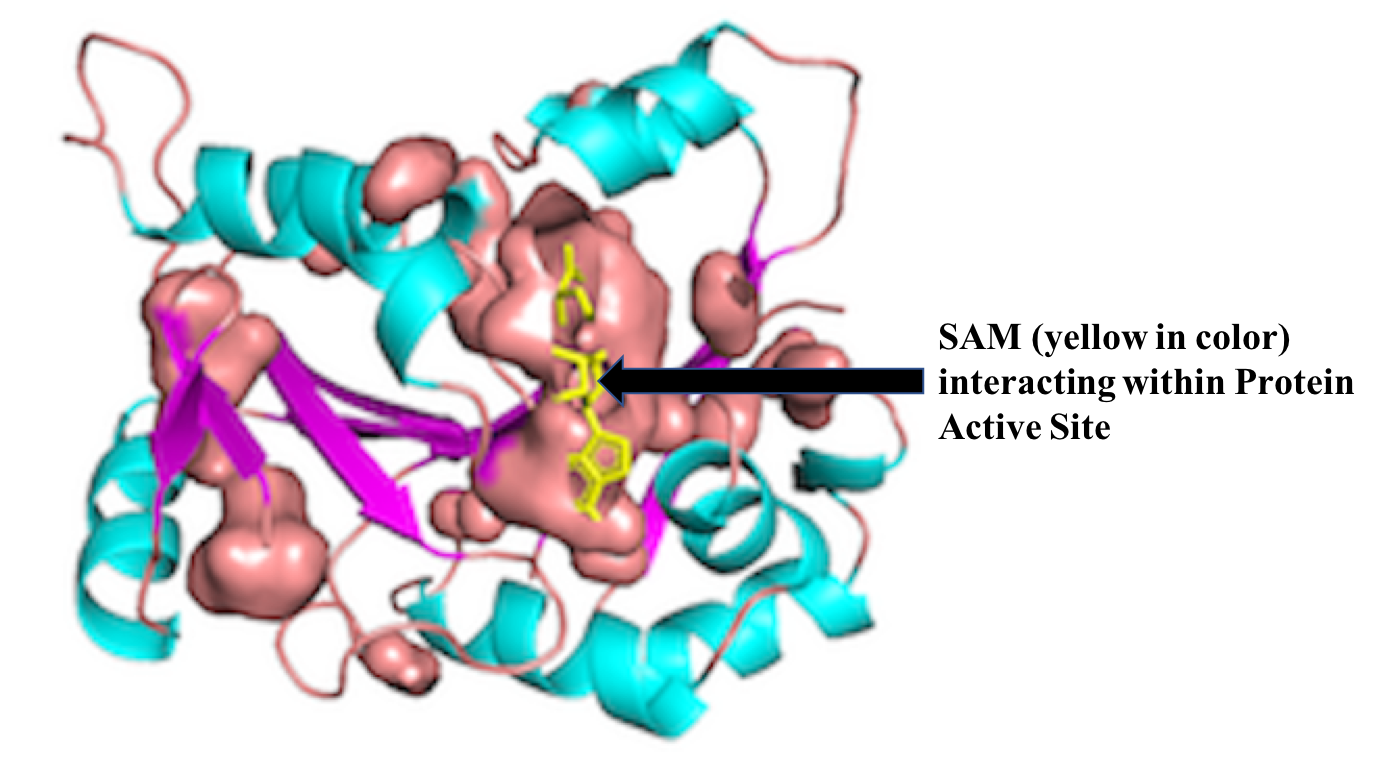
S-adenosyl-L-methionine (SAM) is one of the essential metabolic cofactor/intermediate which is found in almost every cellular life forms and enzymes. It plays a vital role in various metabolic and regulatory pathways, mainly involved in transfer of various groups (e.g., methyl, aminopropyl, methylene). It is a second widely studied ligand after ATP. SAM is a potential drug target for many diseases such as cancer, Alzheimer's, Parkinson, Epilepsy, Osrteoarthritis and many more. Some of the reported drug targets are Catechol O-methyltransferase, Glycine N-methyltransferase, S-adenosylmethionine synthase isoform type-1, S-adenosylmethionine decarboxylase proenzyme, Cystathionine beta-synthase.
SAMbinder is a web server developed for predicting SAM interacting residues in a protein from its primary sequence. This server has following three modules; Sequence based module;PSSM based Module; Peptide Mapping.
Standalone version of SAMbinder has been developed using Python to facilitate user to predict SAM binding sites in proteins on their local machine.
SAMbinder Major Features
- Sequence based module:This module predict SAM binding residues in a protein from its promary sequence.
- PSSM based Module: This module utilize evolutionary information (in form of PSSM profile) of a protein to predict SAM binding residues in a protein.
- Peptide Mapping: This server allow user to predict SAM binding residues in a protein by mapping known peptides that contains SAM interacting central residue.
- Standalone Software: It has been developed using Python and will be freely available to users.
- Datasets: All datasets used in this study will be available to public.
*The server has been checked in different browsers and is best viewed in Safari and Google Chrome.